Long-term, high-resolution intravital imaging of cells and tissues is essential for understanding immune responses, neural activity, and cellular dynamics. However, in thick tissues or complex biological environments, spatially heterogeneous refractive index distributions introduce significant optical aberrations during fluorescence propagation, leading to image blurring and structural distortion. Existing adaptive optics approaches remain limited in accuracy, stability, or system complexity, particularly under conditions of large aberrations or low signal-to-noise ratios. Therefore, developing an aberration correction method that combines high precision with strong robustness is critical for advancing intravital microscopy.
To address these challenges, a team led by Qionghai Dai, Jiamin Wu, and Zhi Lu at Tsinghua University developed a latent-space-enhanced digital adaptive optics method (LEAO). By integrating spatial-angular data from light-field microscopy with deep learning, LEAO enables stable estimation of complex aberrations. Specifically, an autoencoder maps high-dimensional light-field measurements into a latent space, where latent features encoding aberration wavefronts and sample structures are extracted and disentangled. An estimator then predicts continuous wavefront distributions from the aberration latent features, which are used to generate point spread functions consistent with wave-optics models, ultimately improving three-dimensional reconstruction quality. The distilled aberration representation significantly enhances both efficiency and accuracy of wavefront estimation, while disentanglement reduces interference from structural variations.
In systematic evaluations, LEAO maintains stable performance across large aberrations spanning up to five wavelengths and demonstrates strong robustness under low signal-to-noise conditions (3.4 dB). It also generalizes well across varying angle numbers, spatial resolutions, and multiple light-field microscopy configurations. In diverse in vivo experiments, including mouse lymph nodes, cortex, and intact-skull imaging, LEAO consistently recovers clear cellular structures and supports large-scale dynamic analysis. For example, in lymph node imaging, it enables continuous tracking of ~5,000 immune cells over a millimeter-scale field of view. In brain imaging, it increases the number of identifiable neurons and improves signal extraction. Under intact-skull conditions, it corrects severe aberrations introduced by the skull and surgical materials, enabling over 10 hours of dynamic observation of neutrophil activation, extravasation, and clearance following traumatic brain injury.
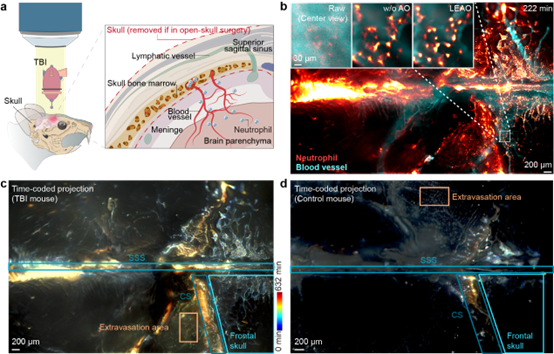
Figure 1. LEAO enables long-term, high-fidelity monitoring of inflammatory responses through the intact mouse skull.
At the methodological level, LEAO introduces a new paradigm that integrates wave-optics modeling with deep learning representations, shifting aberration estimation from explicit modeling toward latent-space-based physical encoding. This strategy provides improved stability and adaptability under complex imaging conditions, offering a powerful tool for multiscale intravital observation. The method holds promise for applications in immunology and neuroscience and may inform the development of next-generation high-fidelity microscopy systems.
The corresponding authors are Qionghai Dai (Academician, Tsinghua University), Jiamin Wu (Associate Professor), and Zhi Lu (Assistant Professor). Yunmin Zeng, a Ph.D. student, is the first author. The team has conducted sustained research in light-field microscopy, adaptive optics, and computational imaging, advancing their applications in life sciences.